
Normalcy vs. time: a brain-cancer patient’s choice
Would you consider a therapy that might get you a few more months, but involved shaving your head and wearing a skullcap-like device for 18 hours a day?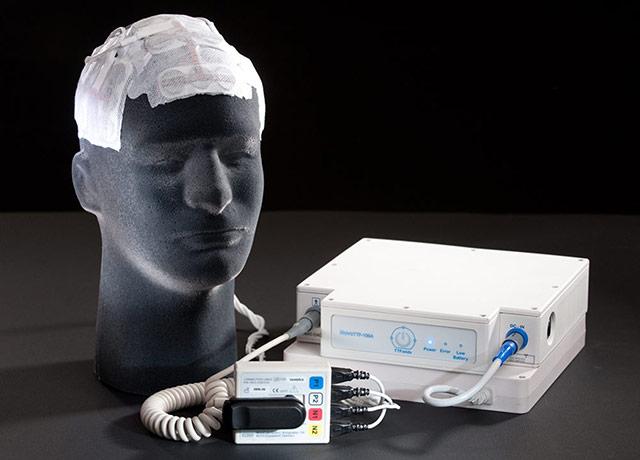
If you were diagnosed with glioblastoma — a brain cancer that, with conventional chemotherapy and radiation treatment, is usually fatal within 12 to 15 months — would you try a novel therapy that might buy you an additional five months (on average)?
What if that therapy involved shaving your head and wearing a skullcap-like array of electrodes for 18 hours a day? Is that worth the possibility of a few more months of life to you?
JAMA Oncology last week published findings from a quality of life survey of brain-cancer patients. The patients participated in a randomized study testing “tumor-treating fields,” the clinical name for the electrode therapy. Dr. Lia Halasz, a UW Medicine radiation oncologist at the Alvord Brain Tumor Center, was not involved in the study but was asked to co-write an accompanying commentary about patients’ decisions to use, or forgo, tumor-treating fields.

Trial results published in December established tumor-treating fields as an important therapy for glioblastoma, Halasz said, gaining patients an average of 4.9 months’ survival time. The array generates electromagnetic fields that interrupt the cell-division process that allows cancer cells to proliferate.
“Many, if not most, of my patients opt against wearing the device, for reasons of comfort and appearance,” she said. “You have to shave your head, which for many people is a big deal. You have to wear a very visible device on your head and you have to carry a battery pack that might affect your activities.
“Patients with cancer want to get on with their daily lives, and with some sense of normalcy. Wearing a device on your head is a constant reminder that you have a glioblastoma.” Halasz likened it to feelings experienced by women whose hair loss from treatment for breast cancer is a constant reminder of their cancer.
In the study, glioblastoma patients who wore the device and those who did not reported similar scores on quality-of-life questionnaires. The only difference was that patients who wore the device had more itchy skin, a known side effect. The finding challenged the study’s hypothesis, Halasz said, which anticipated that array-wearing patients would report worse social interactions and more difficulty with daily physical activities.
“The caveat, of course, is that the patients who were in the trial were open to wearing the device; they elected to participate in the trial, which randomized them to the device or no device.”
“Many patients would opt not to wear it in the first place,” Halasz said. “Some patients are open to it because it’s new and different. But most don’t want the reminder of what they’re dealing with.
“I think this might shift, though, as wearable devices become more prevalent and society grows more comfortable with them.”
For details about UW Medicine, please see our About page.