
New test might expand ability to find tumor cells in blood
Studying tumor cells circulating in the blood makes it possible to track changes in cancer cells and optimize treatment.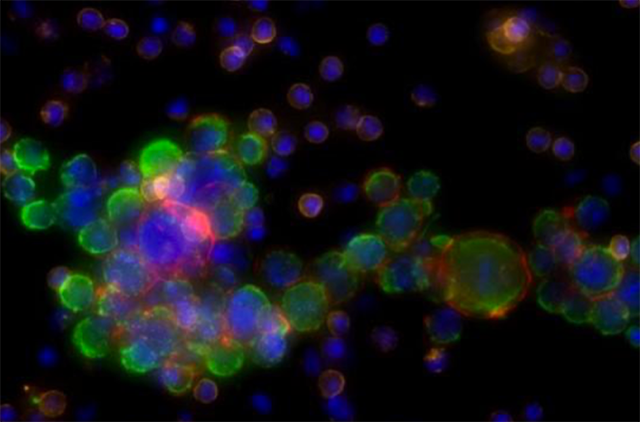
A new blood test that can quantify tumor cells circulating in the bloodstream is ready for testing in the clinic. In preliminary work, the test has been shown to produce results comparable to the only similar test currently approved by the U.S. Food and Drug Administration, researchers in Seattle report.
Both tests detect tumor cells that metastatic cancers shed into the bloodstream, but the new test will be able to detect cells from many more different types of cancer, said Dr. Daniel Sabath, professor of laboratory medicine and pathology at the University of Washington School of Medicine. He developed the new test in collaboration with the Seattle biotech company RareCyte, Inc.
“Circulating tumor cells tell you about a patient’s prognosis,” Sabath said. “If the number of circulating tumor cells in the blood of a patient with metastatic cancer is going up, it indicates their tumor is not responding to treatment. If the number is going down, it suggests the tumor is responding.”
Circulating tumor cells can also be studied to see if a cancer has evolved into a more aggressive form and to identify changes that might make them more vulnerable to other treatments, he said.
Currently, the only FDA-approved circulating tumor cell test is called CELLSEARCH. This test uses magnetic particles coated with antibodies that bind to a protein called epithelial cell adhesion molecule, or EpCam. When the antibody-coated particles are added to a blood sample, a magnetic field can be used to draw out any EpCam-bearing tumor cells. EpCam is found on cells from several different types of tumors, such as lung, breast and colon cancers.

While effective, the test is able to detect only tumor cells that bear EpCam. Some cancers, such as melanoma, do not. Moreover, some EpCam-bearing tumors stop producing the protein as they evolve, thereby allowing them to elude detection.
The test developed by Sabath and his UW Medicine colleagues, in collaboration with RareCyte, takes advantage of the fact that blood components have different densities. That means if they are spun at high speed in a centrifuge tube, they will separate into layers with the heavier components settling towards the bottom and the lighter components floating up towards the top.
To make isolation of the circulating tumor cells easier, the new test uses a centrifuge tube that has a cylindrical float inserted into it that is the same density of the typical circulating tumor cells. As a result, when the tube is spun, red blood cells end up below the float and platelets end up above it. Meanwhile, white cells and tumor cells, which have about the same density, end up trapped in a thin layer of fluid between the cylinder and the wall of the tube. This fluid can then be collected, and the cells put on slides, stained and examined with an automated computer-aided microscopy system
The technique is highly sensitive, according to Sabbath.
“If there is one cell in a sample, we can find it," he said.
In the new study, which appeared in the American Journal of Clinical Pathology Sabath and his colleagues demonstrated in a head-to-head comparison that the RareCyte test performed just as well as CELLSEARCH in detecting breast and prostate cancer cells.
Now that the effectiveness of their technique has been demonstrated, Sabath and his colleagues hope to begin working with researchers conducting clinical trials to show the test can be used to guide and improve treatment.
“Up until now, this has been a research tool, but we want to plug it into clinical trials and show how it can improve patient care,” Sabath said.
Funding from RareCyte and the Department of Laboratory Medicine and Pathology at the UW School of Medicine supported work on the new test.
This news item was written by Michael McCarthy.
Media contact: Leila Gray, 206-475-9809, leilag@uw.edu
For details about UW Medicine, please see our About page.