
Harborview, Airlift Northwest study in-flight transfusion
In clinical trial, some trauma victims will receive type O whole blood instead of red blood cells and plasma, the current standard care.Media Contact: Susan Gregg: 206-616-6730, sghanson@uw.edu
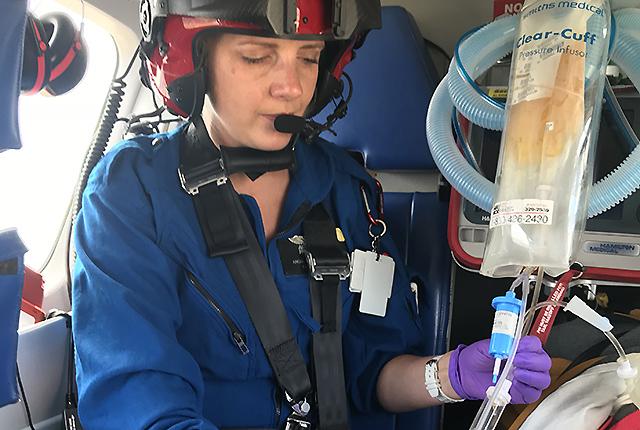
University of Washington School of Medicine researchers are participating in a clinical trial to understand whether trauma victims would benefit from a transfusion of whole blood during prehospital resuscitation. In the study, patients in hemorrhagic shock will be randomized to receive type O-positive low-titer whole blood instead of current standard therapy, red blood cells and plasma, during air transport to the hospital.
“Airlift Northwest has carried red blood cells and plasma on every flight for several years now. Early transfusion of these blood components before arriving at a trauma center has been shown to improve patient outcomes in patients suffering from hemorrhagic shock. Using whole blood in this population has the potential to improve outcomes even further,” said Dr. Richard B. Utarnachitt, medical director at Airlift Northwest and a principal investigator for the Seattle trial site.

UW Medicine colleagues involved in the study are Drs. Andrew Latimer, an emergency medicine specialist at Airlift Northwest, and Bryce Robinson, a critical care specialist at Harborview.
The main trial site is the University of Pittsburgh, where the lead investigator is Dr. Jason Sperry, a trauma surgeon and professor of surgery.
“When someone donates blood, typically it gets separated it into parts—red cells, plasma, and platelets—for storage. The parts can be used individually. But when someone is bleeding, they’re losing all of these parts,” Sperry said. “Trauma research has shown that if you put all of these parts back together again, it can be beneficial to trauma patients or patients at risk of bleeding.”
The whole blood to be used in the study contains low levels of antibodies and can be transfused into people with other blood types. Giving whole blood transfusions to these patients during transport to the hospital might reduce their mortality rate.
"We are excited to be at the cutting edge of the science of treating severely injured patients in the prehospital setting. This has the potential to make a big difference for the sickest patients that we care for and fits well with Airlift Northwest’s mission," adds Latimer.
The trial will begin mid-2022 and is funded by the U.S. Department of Defense. The DOD plans to use the findings of the study to better inform prehospital trauma care for soldiers injured in combat.
The trial investigators hope to answer three key questions:
- Does giving trauma patients whole blood in a prehospital environment improve survival outcomes at 30 days when compared to plasma and red blood cells, or nothing at all?
- Does the age of the whole blood unit affect patients’ survival rates?
- Does administering whole blood provide overall benefit to patients upon evaluation and arrival at the hospital?
This four-year study will involve randomly selected trauma patients who are receiving medical care for significant blood loss. Since patients in such a life-threatening circumstance may be unable to give informed consent to be involved in the study, the researchers have acquired a federal exception that allows them to enroll patients without consent. However, patients who receive whole blood may opt out of study follow-up during their recovery, and people also can opt out of the trial in advance by calling 800-664-0557 or completing this online form.
This research is supported by Department of Defense contract W81XWH-16-D-0024 W81XWH18F0426. Any opinions, findings and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the Department of the Army, Department of Defense, or the U.S. Government.
For details about UW Medicine, please see our About page.
Topics:informed consentplasma