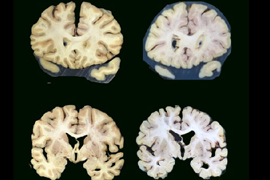
Traumatic brain injury raises risk of brain atrophy
Study adds to other findings suggesting pathology of post-traumatic brain cell loss is distinct from Alzheimer’s disease.Media Contact: Susan Gregg - sghanson@uw.edu, 206.390.3226
It has long been known that people who have had traumatic brain injury with loss of consciousness have an increased risk of dementia. But it has been debated whether the increased risk was due to brain changes like those seen with Alzheimer’s disease and related dementias or some other process.
Now a new autopsy study from researchers at the University of Washington School of Medicine, Mount Sinai School of Medicine, and Kaiser Permanente Health Research Institute has found that people who reported having lost consciousness after sustaining a traumatic brain injury faced a higher risk of brain atrophy, but not the changes associated with Alzheimer’s and related dementias. Brain atrophy is a loss of brain nerve cells and the connections between them. This often leads to decreased brain volume.
“Our finding adds to a growing body of research that suggests the pathological processes underlying post-traumatic neurodegeneration are distinct from those seen with Alzheimer’s disease,” said Laura Gibbons, a senior research scientist at the UW School of Medicine’s Department of Internal Medicine and lead author on the study. The article was published online in the Journal of Alzheimer’s Disease in April.
In their study, the UW Medicine researchers and their colleagues compared the brains of people who reported having had head trauma with loss of consciousness with those of people who reported never having such an injury. The brains were donated by participants in a Seattle study of brain aging and dementia called the Adult Changes in Thought study. Participants in the ACT study were members of Kaiser Permanente Washington in the Seattle area who were randomly selected to be invited to participate. To qualify, they had to be 65 or older, living in the community and not have dementia.
At the time of enrollment and every two years thereafter, the participants underwent detailed health and cognitive assessments until they either died, developed dementia or dropped out. As part of their assessments, information was gathered from the participants, their medical records and, in the event of their death or the onset of dementia, from interviews with family and friends. The focus was the participant’s history of head trauma, including whether it was accompanied by loss of consciousness, which can occur with more severe head trauma.
Examination of the participants' donated brains did not find more of the changes seen with Alzheimer’s disease and related dementias, such as amyloid plaques, in those who had brain injury with loss of consciousness. But they did find that those who had experienced head trauma with loss of consciousness were more likely to have brain atrophy.
The increase in risk for brain atrophy was small, or about 22%, ”but it was statistically significant, so it’s likely real," noted C. Dirk Keene, the Nancy and Buster Alvord Endowed Chair in Neuropathology and professor of laboratory medicine and pathology at the UW School of Medicine, whose team examined the brains. The cause of brain atrophy in these individuals is unknown, Keene said.
“We typically see atrophy as the downstream consequence of many other forms of neuropathology,” said Keene. “Strokes can cause atrophy, Alzheimer's-type neuropathological changes like amyloid plaques can cause atrophy, and so on. But we looked for everything else and did not find any other clear associations with TBI—just atrophy itself. Atrophy is also a consequence of the diffuse injury to axons commonly seen after brain injury, so this finding supports axonal injury as a contributor to post-traumatic neurodegeneration. We still have a lot to figure out."
The research was funded by the National Institute on Aging (U19AG066567, U01AG006781, P30 AG066509, pP50AG05136, AG0610280), the National Institutes of Neurological Disorders and Stroke (U01NS086625-01), the Department of Defense Congressionally Directed Medical Research Program (W81XWH-17-1-0330), and the Nancy and Buster Alvord Endowment.
This news item was written by Michael McCarthy.
Among people whose liver is failing, the perpetual shortage of donor organs inhibits expectations of a timely, life-saving transplant. New research suggests that these people who initially seek care at safety-net hospitals may face additional obstacles to being considered for transplant.
A study of three safety-net hospitals showed that, among patients whose measures of liver health would typically result in a referral for transplant evaluation, only about one-fourth received the referral. The finding was published June 8 in JAMA Network Open.
A safety-net hospital has a mission or a legal obligation to provide healthcare to individuals regardless of their ability to pay for it. In the study, each of the three hospitals — LA County and Zuckerberg San Francisco General, both in California, and Harborview Medical Center in Seattle — is affiliated with another medical center that has an active, well-regarded transplant program.
“Patients who receive care at these safety-net hospitals generally do not go to the other hospital unless they have a specific referral,” said Dr. Nicole Kim, a co-author on the paper and a liver transplant fellow at the University of Washington School of Medicine. “Patients who present at safety-net hospitals are often more likely to identify as indigent and underserved, uninsured, lower-income, racial-ethnic minority and-or homeless.”
Although someone’s socioeconomic status might not directly affect their MELD (Model for End Stage Liver Disease) score predicting survival in people with liver disease, a patient’s apparent lack of social support and their perceived inability to pay for a lifetime of anti-organ rejection meds could influence a doctor’s decision about referral, Kim said.
“It’s knowing that for a transplant to be successful, which is the end goal of everyone, the patient must have a support group of people to provide care as they’re recovering,” Kim said. “They also need to be able to acquire medications for the rest of their lives.”
The retrospective study comprised 521 patients who were seen at the three safety-net hospital liver clinics in 2016-17 and followed until 2019. All patients had received a formal diagnosis of cirrhosis, meaning irreversible scarring of the liver, and all had MELD scores of 15 points or greater, “which is routinely the cutoff at which we’d tell a patient, ‘Your liver is sick enough that you may want to consider transplant,’” Kim said.
Of the 521 patients, 145 (27.8%) were referred to a transplant specialist for evaluation — a percentage Kim characterized as “quite low.” The study did not comparatively review the rate of referrals from non-safety-net hospitals among patients with similar stage of liver disease.
The researchers identified three common factors among the patients who did not receive referrals for transplant evaluation: undocumented citizenship, unstable housing and uninsured status. The study identified these traits as “barriers” to referral.
“I think a lot depends on the provider knowing that the patient doesn’t have insurance and-or likely will have difficulty adhering to the medical requirements that accompany transplant, and as a result might tell someone, ‘Maybe now is not the right time for a referral,’” Kim said, who has cared for patients at Harborview’s Liver Clinic.
The findings highlight the need, Kim said, for safety-net hospitals to better identify patients’ eligibility for insurance or housing that would make them a more viable candidate for transplant — and then help navigate them to those potential resources.
“There are always going to be patients who are not eligible for insurance, who are not eligible for stable housing, for various reasons. But we're really talking about the folks who may be eligible but don't know,” she said. “These are the folks that should be getting that extra level of support to receive the care they need.”
The study was supported by funding from the National Institute of Diabetes and Digestive and Kidney Diseases (P30 DK026743, T32DK007742), part of the National Institutes of Health.
For details about UW Medicine, please visit https://uwmedicine.org/about.