
FDA aims to level field on breast-density disclosure to patients
The new regulation demands more communication from screening facilities across 12 states, including Alaska, Wyoming, Idaho and Montana.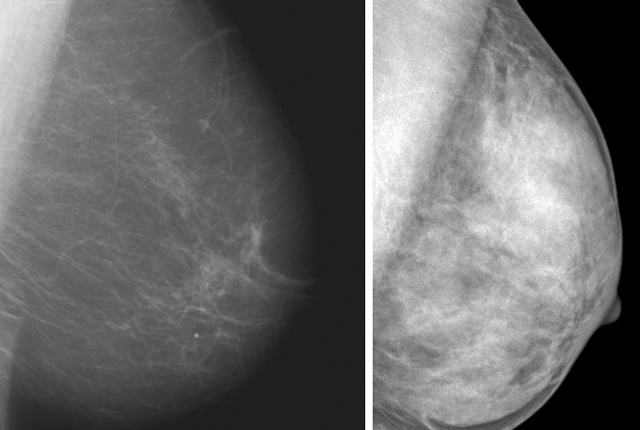
Among six Pacific Northwest states, only Washington and Oregon have laws mandating that radiologists notify female patients whose mammograms indicate dense breast tissue, a risk factor for cancer.
A decision this month by the Food and Drug Administration, however, aims to broaden that awareness. Its requires U.S. mammography facilities to provide personal breast-density information to all patients. This will compel new behavior among clinicians in Alaska, Wyoming, Idaho, Montana and eight other U.S. states that have no such law.
“The intent is to help women by informing them of a breast cancer risk factor they may not be aware of,” said Dr. Christoph Lee, an expert in mammography and a professor of radiology at the University of Washington School of Medicine.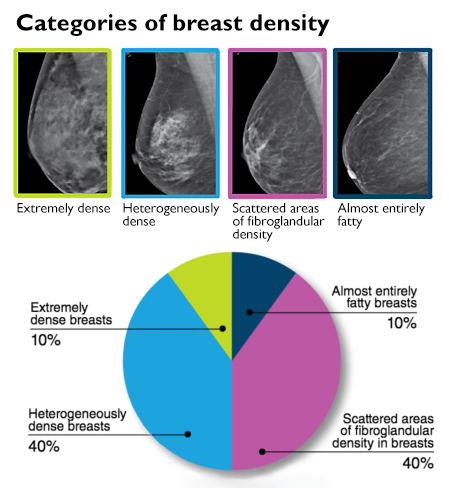
Dense breast tissue is a “moderate” risk factor for cancer, said Lee, who also directs the Northwest Screening and Cancer Outcomes Research Enterprise. Family history of breast cancer and personal history of breast tumors, cancerous or benign, are stronger risk factors than dense breasts, he said.
He explained that radiologists subjectively review mammograms to characterize breast tissue into one of four categories, which range from almost entirely fatty to extremely dense with fibroglandular tissue (see accompanying chart).
It’s important to note, Lee said, that half of all women are classified as having dense breasts just by the nature of the categories.
The new FDA regs dictate that women must be told whether their breasts are “dense” or “non-dense,” but do not require the direct disclosure of their more specific tissue category. That information must be shared with the patient’s primary care physician, though, to inform a doctor-patient conversation that Lee says is useful for women with dense breasts.
“The FDA rule asks that physicians and patients have an informed discussion and share that decision-making about how to use that risk information,” he said.
At that point, the main question is whether a patient needs additional tests beyond mammography. The majority of women with dense breasts do not need additional tests, Lee said.
“For those women with extremely dense breasts, which are only 10% of the population getting screened, it may be appropriate to add a supplemental screening test if they have other risk factors,” he added.
Additional screenings would involve ultrasound and/or MRI, technologies less susceptible to having tumors obscured by dense tissue.
Written by Brian Donohue - 206-543-7856, bdonohue@uw.edu
[Download accompanying video soundbites with Dr. Christoph Lee.]
For details about UW Medicine, please see our About page.