
Delving into the 'why' of urinary stent pain
Some patients experience agonizing pain when a temporary stent is implanted after kidney stones are removed.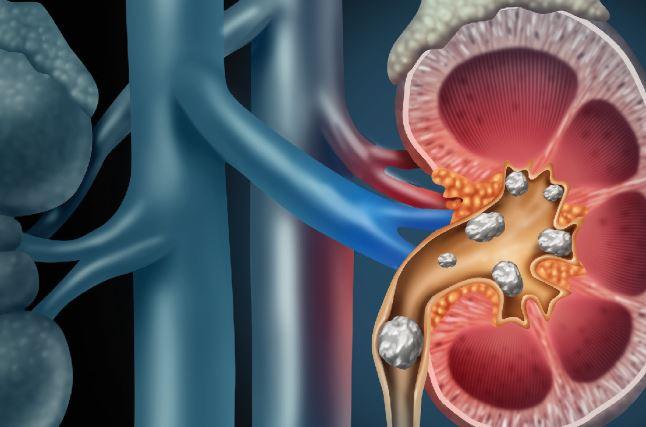
For most, getting kidney stones removed through an ureteroscopy might be an uncomfortable but tolerable procedure.
For about 20 percent of patients, though, the procedure of treating a stone and placing a temporary ureteral stent – to counteract swelling and allow passage of urine from the kidney to the bladder - is agonizing. A study being conducted at UW Medicine’s Department of Urology will examine why.
Ureteroscopy is a procedure in which a small scope is inserted into the urethra and advanced to the bladder and kidneys. It allows the urologist to find the stone and remove it. Once the procedure is complete, the doctor often places a temporary stent in the ureter, between the kidney and the bladder; this helps to drain small residual fragments and is thought to help the ureter heal, said Jonathan Harper, a urologist at the University of Washington School of Medicine.
[Downloadable video soundbites of Jonathan Harper discussing the stent study]
“For some, the placement (of the stent) is almost intolerable. We don’t fully understand who is at risk for severe symptoms, what triggers them, or why," Harper said.
He and colleagues at a few other medical centers aim to enroll 400 patients over the next year. The first 40 enrollees will undergo a more extensive evaluation about their experiences. They will complete daily questionnaires about urinary and pain symptoms and undergo pain sensory testing. The researchers hope the resulting information will help them predict which patients will not tolerate a stent.
“If we can identify patients at high risk for severe symptoms and emergency room visits, we could then study the use of targeted medications or behavioral counseling to deal with the pain in future clinical trials,” Harper said.
During the study, urine and blood samples will be collected to establish a national biorepository. Researchers hope this data bank will eventually identify inflammatory biomarkers so doctors can predict which patients will react adversely to stents.
Interested patients can find more information here.
This project is funded by the the National Institute of Diabetes, Digestive and Kidney Diseases and the Urinary Stone Disease Research Network.
Media contact: Barbara Clements, bac60@uw.edu, 206-221-6706.
For details about UW Medicine, please see our About page.
Topics:kidney stones