
Scientists create complex transmembrane proteins from scratch
The ability to build transmembrane proteins opens the way for custom-designing structures that span living cell membranes and perform new tasks.Media Contact: Leila Gray, 206.685.0381, leilag@uw.edu
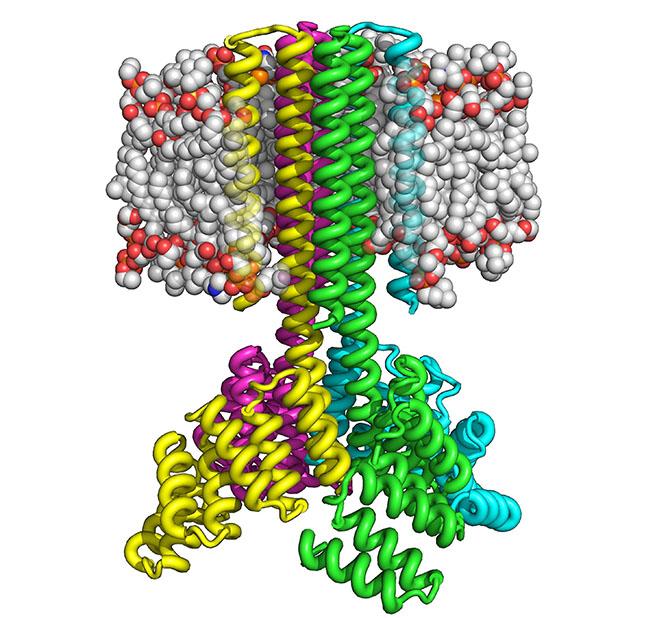
In the living world, transmembrane proteins are found embedded in the membrane of all cells and cellular organelles. They are essential for them to function normally. For example, many naturally occurring transmembrane proteins act as gateways for the movement of specific substances across a biological membrane. Some transmembrane proteins receive or transmit cell signals. Because of such roles, many drugs are designed to target transmembrane proteins and alter their function.
Now researcher are looking at designing the transmembrane proteins themselve to perform specific tasks.
"Our results pave the way for the design of multispan membrane proteins that could mimic proteins found in nature or have entirely novel structure, function and uses," said David Baker, a University of Washington School of Medicine professor biochemistry and director of the UW Institute of Protein Design who led the project. The research is reported in the March 1 issue of the journal Science. Peilong Lu, a senior fellow in the Baker lab, is the paper's lead author.
But understanding how transmembrane proteins are put together and how they work has proved challenging. Because they act while embedded within the cellular membrane, transmembrane proteins have proven to be more difficult to study than proteins that operate in the watery solution that make up the cells' cytoplasm or in the extracellular fluid.
In the new study, Lu and his coworkers used a computer program, developed in the Baker lab and called Rosetta, that can predict the structure a protein will fold into after it has been synthesized. The architecture of a protein is crucial because a protein's structure determines its function.
A protein's shape forms from complex interactions between the amino acids that make up the protein chain and between the amino acids and the surrounding environment. Ultimately, the protein assumes the shape that best balances out all these factors so that the protein achieves the lowest possible energy state.
The Rosetta program used by Lu and his colleagues can predict the structure of a protein by taking into account these interactions and calculating the lowest overall energy state. It is not unusual for the program to create tens of thousands of model structures for an amino acid sequence and then identify the ones with lowest energy state. The resulting models have been shown to accurately represent the structure the sequence will likely assume in nature.
Determining the structure of transmembrane proteins is difficult because portions of transmembrane proteins must pass though the membrane's interior, which is made of oily fats called lipids.
In aqueous fluids, amino acid residues that have polar sidechains - components that can have a charge under certain physiological conditions or that participate in hydrogen bonding -- tend to be located on the surface of the protein where they can interact with water, which has negatively and positively side charges to its molecule. As a result, polar residues on proteins are called hydrophilic, or "water-loving."
Non-polar residues, on the other hand, tend to be found packed within the protein core away from the polar aqueous fluid. Such residues are called hydrophobic or "water-fearing." As a result, the interaction between the water-loving and water-fearing residues of the protein and the surrounding watery fluids helps drive protein folding and stabilizes the protein's final structure.
In membranes, however, protein folding is more complicated because the lipid interior of the membrane is non-polar, that is, it has no separation of electrical charges. This means to be stable the protein must place nonpolar, water-fearing residues on its surface, and pack its polar, water-loving residues inside. Then it must find a way to stabilize its structure by creating bonds between the hydrophilic residues within its core.
The key to solving the problem, said Lu, was to apply a method developed by Baker lab to design the transmembrane portion so that the polar, hydrophilic residues fit in such a way that enough would form hydrongen bonds-- that can tie the protein together from within
"Putting together these 'buried hydrogen bond networks' was like putting together a jig-saw puzzle," Baker said.
With this approach, Lu and his colleagues were able to manufacture the designed transmembrane proteins inside bacteria and mammalian cells by using as many as 215 amino acids. The resulting proteins proved to be highly thermally stable and able to correctly orient themselves on the membrane. Like naturally occurring transmembrane proteins, the proteins are multipass, meaning they traverse the membrane several times, and assemble into stable multi-protein complexes, such as dimers, trimers and tetramers.
"We have shown that it is now possible to accurately design complex, multipass transmembrane proteins that can be expressed in cells. This will make it possible for researchers to design transmembrane proteins with entirely novel structures and functions," said Lu.
This work was supported by the Howard Hughes Medical Institute, National Institutes of Health (R01GM063919), the Raymond and Beverly Sackler fellowship, and the National Research Foundation of Korea (NRF- 2016R1A6A3A03007871).
For details about UW Medicine, please see our About page.
Topics:Institute for Protein Design