
COVID-19 coronavirus spike holds infectivity details
Several new findings on how the virus’ cell entry tools operate could expedite the design of vaccines, antibodies, antivirals and other therapeutics.Media Contact: Leila Gray - 206.685.0381, leilag@uw.edu
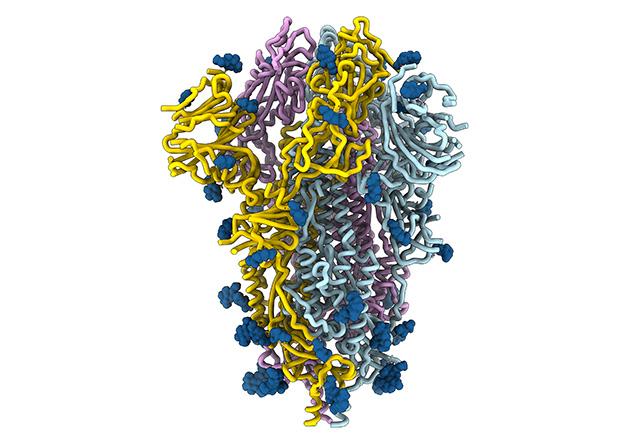
The spikes crowning the new coronavirus that causes COVID-19 atypical pneumonia are divulging how they attach, fuse and gain entry to cells. Analysis of the spike architecture and its mechanics is locating the virus’ vulnerabilities, and revealing other information that could prompt the discovery of countermeasures against this virus.
A research team at the University of Washington School of Medicine and Fred Hutchinson Cancer Research Institute uses cryo-electron microscopy and other investigative methods in this effort. They are helping to determine the structure and function of the SARS-CoV-2 spike protein and its chemical binding affinities as these relate to both infection and immune responses, and thereby obtain ideas for blocking the virus’ ingress to cells.
Their multiple findings were published as a preliminary report Feb.20 in bioRxiv, a preprint server for biology. The lead authors are Alexandra C. “Lexi” Walls, a recent postdoctoral fellow, and Young-Jun Park, a research scientist. Both conduct coronavirus studies in the lab of David Veesler, senior author of the report and assistant professor of biochemistry at the UW School of Medicine.
“The spike is the business part as far as viral entry is concerned,” Veesler explained. “It is in charge not only of attachment at the host cell surface, but also of fusing the viral and host cell membranes to allow the infection to start. The spike is also the main target of neutralizing antibodies, so it’s very important for vaccine and therapeutic design.”
While the Seattle researchers have been tirelessly examining the spike structure and function of new coronavirus since shortly after the disease outbreak, over the past several years they have performed similar spike studies of other serious coronaviruses, including the types that cause the diseases SARS (Severe Acute Respiratory Syndrome) and MERS (Middle East Respiratory Syndrome). They also study attachment and fusion proteins in the bat-harbored Hendra and Nipah henipaviruses.
One finding in their recent research was the detection of cross-neutralizing antibodies that inhibit the cellular fusion by SARS-CoV and SARS-CoV-2, and that these antibodies might be elicited by vaccination.
“The fact that antibodies elicited by the spike proteins in related SARS coronaviruses also neutralized infection with this new one is an important step forward to find antibodies and design vaccines that could block this group of coronaviruses,” Veesler said. These could provide broad protection against several SARS-CoV-like pathogens.
His group also showed that the cell surface receptor, angiotensin converting enzyme 2 (ACE2), is recognized by the new coronavirus and serves as the access point into human cells. This is also the receptor for SARS-CoV. The research results are in agreement with some similar, recently reported findings by other scientific teams demonstrating human ACE2 is a functional receptor for the new virus.
Previous work suggested that spike protein adaptation that causes a high affinity for human ACE2 receptors may be related to the severity of SARS-like coronavirus disease. The team showed that SARS-CoV-2 binds at least as well as SARS-CoV to ACE2. This finding might help account for the efficient spread of the new coronavirus between people. As with several other coronaviruses, the new virus probably originated in an animal which remains its reservoir, most likely a species of bat. Then, possibly through a different intermediary animal species, the virus jumped to humans, and became able to be transmitted between people. The exact origin of the new coronavirus disease has not yet been firmly established.
By closely examining the structure of the new coronavirus spike protein, the researchers did uncover something that sets it apart from the other SARS-related coronaviruses. Walls said that the research team unexpectedly found a furin cleavage site at a boundary between two subunits of the spike protein in the newly emerged coronavirus. It is not yet known if this difference is expanding the kinds of cells the new coronavirus could infect or enhancing its transmissibility, in a way that might be similar to that of highly pathogenic avian flu viruses.
As a whole, details contained in the results reported in this week’s paper may help to explain the efficiency of the new coronavirus in delivering its viral code into human cells, and its rapid transmission among people.

At present, there are no preventatives or approved, specific treatments (aside from experimental therapies) that can be directed at the new coronavirus.
“Our ultimate goal would be if our work could contribute as a step towards a vaccine, antiviral or any sort of therapy that currently does not yet exist,” Walls said, and added that it would be fantastic if her team’s scientific efforts could be a step towards being helpful to people in that way.
Veesler described Walls and Park and others on the team as “amazing scientists who have been working almost 24 hours a day in the lab over the past month to obtain these amazing results” to contribute to advancing the field of coronavirus research and to addressing the current public health emergency .
Parks acknowledged that the hours have been long, but that didn’t matter because he is motivated in the lab by what patients in the midst of the epidemic have had to face.
“This is what we and other labs are doing to combat this virus, and the emergence and spread of future coronaviruses that might happen, as might be the case for other viruses found in wildlife. We should just work to be prepared for it,” Veesler said.
This study was supported by the National Institutes of General Medical Sciences (R01GM120553), National Institutes of Allergy and Infectious Diseases (HHSN272201700059C), a Pew Biomedical Scholars Award, and Investigators in the Pathogenesis of Infectious Disease Award from the Burroughs Wellcome Fund, the Open Philanthropic Foundation, and the Pasteur Institute.
The researchers declared that they have no competing financial interests.
For details about UW Medicine, please visit https://uwmedicine.org/about.