
Brain scans suggest subtypes of Alzheimer’s disease
The findings support the theory that there might be biologically distinct forms of the disease.Media Contact: Leila Gray, 206-475-9809, leilag@uw.edu
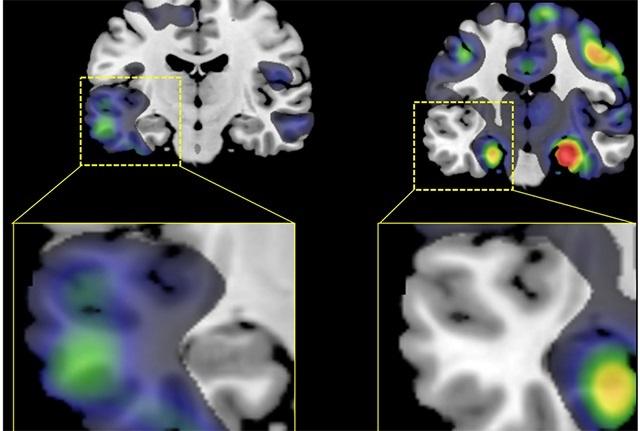
Brain scans have shown marked anatomical differences among the brains of patients with different subtypes of Alzheimer’s disease. Researchers from the United States and Europe report these findings in a paper published Dec. 13 in the journal Alzheimer’s & Dementia.
The new evidence supports the theory that there are multiple, biologically distinct subgroups of the disease. If this is correct, it suggests that different disease factors could be driving the different subtypes, and that distinct treatments targeting those factors could improve individuals’ care.
“Breast cancer was once considered one disease,” said lead author Dr. Paul Crane, professor of medicine, Division of General Internal Medicine, at the University of Washington School of Medicine in Seattle. “But we now know there are different subtypes of breast cancer that respond to different treatments. Our findings suggest a similar approach may apply to Alzheimer’s.”
In the study, Crane and colleagues analyzed data from magnetic resonance imaging of the brains of 584 patients with Alzheimer’s. The controls were 118 people without the disease. The participants, who were 65 or older, were scanned when they were first diagnosed with the disease.
The MRI data came from the Alzheimer’s Disease Neuroimaging Initiative, an international collaborative that seeks to better diagnose Alzheimer’s early and track its progression in hopes of developing ways to prevent and treat the disease.
All the patients had undergone a battery of cognitive tests. Their results enabled the researchers to assign them to subgroups of Alzheimer’s disease based on the cognitive functions most severely affected.
“We compared each person’s scores for four domains – memory, executive functioning, language and visuospatial,” Crane explained. “Most people with Alzheimer’s have difficulties with these domains which are fairly consistent across the board. But some people have particularly marked difficulties for one domain. We define people in those groups as having AD-Memory, AD-Executive functioning, AD-Language, and AD-Visuospatial, all based on the pattern of findings at the time they were first diagnosed with Alzheimer’s.”
The researchers found that the volume of gray matter was lower — a sign of atrophy — in different regions of the brain depending on the subtype of the disease afflicting the patient. Those in the AD-Memory subtype, for example, had lower volumes in an area known to be crucial for learning and memory, called the medial temporal lobe, while the rest of the brain was relatively spared. Those in the AD-Visuospatial subtype, on the other hand, had lower volume in the gray matter toward the back of the head, where visual information is processed. Those with the AD-Executive functioning subtype had lower volume in the front of the brain, in areas where planning, judgement and other higher-order functions are known to reside.
Of particular interest was the finding that lower volumes were strikingly asymmetrical in patients with the AD-Language subtype compared with those in any other subgroup or to the normal controls. In these patients, atrophy was notably greater on the left side of the brain than on the right.
“Alzheimer’s has long been considered a symmetrical disease, so pathological studies have often been based on samples taken from just one side of the brain,” Crane noted. “As a result, it’s possible we’re missing important pathology that might be occurring on the other side of the brain.
“The results of this study strengthen the evidence from clinical, genetic and previous imaging studies that there are distinct subtypes of Alzheimer’s disease and that these subtypes have biologically meaningful differences,” he continued. “This raises the possibility that Alzheimer’s therapy might be personalized to treat patients depending on the subtype of their disease.”
Support for this research came from the National Institutes of Health (R01AG 029672, U01 AG024904), the U.S. Department of Defense (W81XWH-12-2-0012), National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, the Alzheimer’s Association and the Alzheimer’s Drug Discovery Foundation. The Alzheimer’s Disease Neuroimaging Initiative is a public-private partnership supported with industry, foundation and government funds.
For details about UW Medicine, please visit https://uwmedicine.org/about.