
Atomic-level study captures frog toxin in action
The unexpected findings reveal why this paralyzing, heart-stopping chemical can be instantaneously lethal.Media Contact: Chris Talbott - 206-543-7129, talbottc@uw.edu
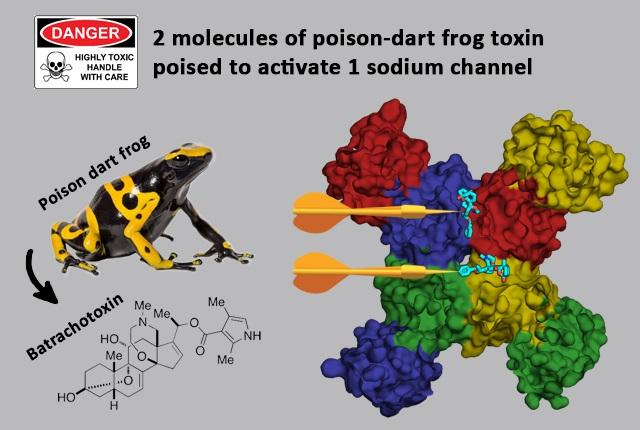
Cryo-electron imaging studies of poison dart frog toxin molecules in action are helping to solve the mystery of why this is among the deadliest natural poisons.
When threatened, some tiny Amazon frogs defend themselves by releasing a substance that quickly incapacitates a predator. For centuries, a few Indigenous tribes in Colombia have siphoned this toxin from skin glands on the frogs’ backs to coat blow darts for hunting, because of its power to take down big game.
The poison instantly cramps the muscles in the body and forces the heart into a chaotic rhythm that ends in cardiac arrest. The bright coloring of the frogs warns animals to stay away. Their chemical weapon is called batrachotoxin, a name based on the Greek root word for frog.
“Predators that attack these dendrobatid frogs are paralyzed so rapidly that they can’t clamp down on the frogs to kill and eat them,” said one of the senior researchers on a recent batrachotoxin study, William Catterall, a professor of pharmacology at the University of Washington School of Medicine.
“It is so toxic that wiping the toxin secretion from the back of a single frog is sufficient to poison several blow darts, which will each immobilize or kill large prey,” he noted.
What makes their irreversible toxin more severe, compared with others that also target the signal-producing pores of nerve and muscle cells?
It has long been known that batrachotoxin affects sodium ion channels, which control electrical pulses by regulating the flow of charged sodium particles through the membranes of these cells.
“The toxin acts by hyperactivation of the sodium channel that generates action potentials in nerve and muscle, thereby causing a lethal storm of hyperactivity of nerves, synapses and muscle cells,” Catterall said.
By using high-resolution cryogenic electron microscopy, a research team led by pharmacologists at the University of Washington School of Medicine have determined, at the atomic level, the structure of the toxin derivative batrachotoxin-A benzoate when it is bound to its receptor site on the cardiac sodium channel.
Unexpectedly, the researchers discovered that, unlike other toxins, batrachotoxin actually binds to two separate but similar receptor sites in the cardiac sodium channel. This dual-receptor action, and its multifaceted effects, provide unprecedented insights into why this poison is so effective, powerful and deadly.
The toxin's unique mechanism for high potency and lethality might also establish a new paradigm that helps to explain the actions of other toxins with high lethality, the researchers suggested.
Such studies might also lead to better therapies for conditions resulting from certain electrical signaling problems in excitable cells and tissues, such as those in the heart, muscles, nerves and brain that are caused by malfunction of a sodium channel.
For example, Catterall suggested, drugs designed to be robust inhibitors at both dual-receptor sites discovered in this study might also be valuable therapeutic agents in the treatment of disorders of hyperexcitability of nerve and heart cells. Such disorders include epilepsy, chronic pain and arrhythmias.
The findings on the poison frog toxin were published March 14 in Nature Communications. The lead authors are Lige Tonggu, Goragot Wisedchaisri and Tamer M. Gamal El-Din, and the senior authors are Catterall and Ning Zheng, all in the Department of Pharmacology at the UW School of Medicine. They collaborated on the work with scientists in the Department of Chemistry at Stanford University.
The research was supported by National Institutes of Health research grants K08 HL145630, R35 NS111573, R01 HL112808 and R01 GM117263. Ning Zheng is a Howard Hughes Medical Institute investigator. A portion of this research was supported by National Institutes of Health Research Grant U24GM129547 and performed at the Pacific Northwest Center for Cryo-EM at Oregon Health & Science University, a U.S. Department of Energy Office of Science User Facility sponsored by the Office of Biological & Environmental Research.
Editor's note: The research team honors the memory of Dr. William A. Catterall (Oct. 12, 1946 – Feb. 28, 2024), the senior author on this paper who was internationally recognized for his pioneering discoveries involving ion channels. His fellow scientists, Ning Zheng and Goragot Wisedchaisri, can answer media questions about this work.
Written by Leila Gray
For details about UW Medicine, please see our About page.