
Prowess, persistence yield first-anywhere heart procedure
Cardiologists detached and removed a patient's failed mitral-valve clip device, then implanted a new biologic valve — all via catheter.Media Contact: Brian Donohue - 206-543-7856, bdonohue@uw.edu
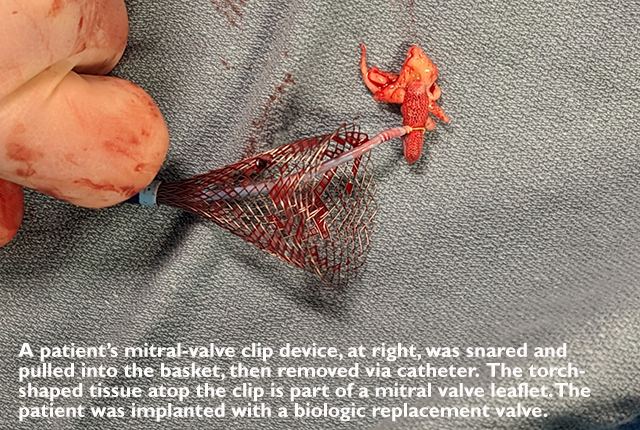
Cardiologists at the UW Medicine Heart Institute recently performed a first-in-the-world procedure, detaching and retrieving a clip device from a patient’s mitral valve and placing a replacement biologic valve — all through a catheter. Previously this sequence of tasks had been performed only via open surgery.
The patient, John Steenmeyer, 76, of Bellingham, Wash., was discharged this week from the University of Washington Medical Center in Seattle. His new mitral valve is functioning well, said Dr. James McCabe, who led the catheterization procedure on April 27.

For years, Steenmeyer’s energy had declined as a poor seal between the two leaflets of his mitral valve had allowed blood to leak backward into the left atrium. Such “regurgitation” is a common heart-valve problem that, in mild cases, someone can live with for years, but in severe cases requires medical intervention.
“It affected my life quite a bit,” Steenmeyer said of his failing valve. “It got to the point that I couldn’t do anything with the cadets,” he said, referencing his role as Whatcom County's commanding officer of the Sea Cadets, a U.S. Navy-sponsored youth development program.
In 2021, his Bellingham cardiologists determined he could wait no longer, but also that surgery was too risky. Steenmeyer underwent a catheter-based procedure to implant a MitraClip, a clothespin-like device that pulled his mitral valve’s two flapping leaflets together to improve their seal. Alas, soon after, his doctors realized that the clip was not sufficiently reducing the leak.
In August 2022, he was referred to UW Medicine. “I was ready to make out my will,” Steenmeyer said, “so this came along just at the right time.”
McCabe said he routinely fields calls from regional cardiologists asking, “‘Hey, we don’t know what to do for this patient. Can you help?’ So we start with a pretty open playbook.”
His first thought was that, as with other patients in this circumstance, Steenmeyer might benefit from an additional MitraClip.

“You can stack them but also you have to be thinking: How small are you making the valve openings? There’s an inflection point at which you can make the valve too small, and then you’re trading leak for stenosis [over-narrowing] and other problems.”
Ultimately, calcium deposits on one side of Steenmeyer’s mitral valve made the decision for McCabe: Since they would continue to expand, and shrink the valve’s circumference, no additional MitraClips could be placed. The subsequent confirmation that Steenmeyer was not surgery-viable seemed to exhaust his options.
McCabe approached Edwards Lifesciences, a company that makes replacement valves. One of its new devices, a catheter-placed mitral valve, is currently being tested in a national clinical trial. McCabe is UW Medicine’s principal investigator in that trial.
He asked Edwards: If there was a way to remove Steenmeyer’s MitraClip without surgery, could he be considered for the test device? The answer was a qualified yes; Steenmeyer couldn’t be included in the trial, but could get the test valve if the U.S. Food and Drug Administration would authorize a Compassionate Use exemption.
Getting the approvals took six months. “During that time, we made a plan that leveraged our experience from last year,” McCabe said, describing another first-anywhere catheter procedure in which electrocautery was used to cut a benign tumor away from a patient’s inner heart wall and remove it in a small basket.
He described Steenmeyer’s procedure: “We cut the back leaflet, and while we’re holding onto the clip, we cut the front leaflet and then drew it back into this basket. We had to pull it from the left atrium across the wall between the two atria, where we’d made a hole, into a large sheath in his interior vena cava, then we removed it.
“We’re just trying to emulate surgery for people who can’t have surgery,” said McCabe. “We’re glad Mr. Steenmeyer is doing well.”
Neither Edwards Lifesciences nor Medtronic, another manufacturer of a transcatheter mitral valve in clinical trial, was aware of a previous procedure involving their device and a catheter-based MitraClip extraction, McCabe confirmed.
McCabe was assisted in the procedure by Drs. Gabriel Aldea and David Elison, and cardiovascular technologist Bailey Benton. Dr. Burkhard Mackensen provided interventional echocardiography.
For details about UW Medicine, please see our About page.